Monoclonal Antibodies: A new era of personalized medicine
Our "one-size fits all" medical model is outdated - are monoclonal antibodies the key for individualized health-care?
No two humans are alike (with only some exceptions). We each have our own molecular differences (i.e. produce more of a particular protein, lack an essential enzyme, possess an oncogene, etc.) that could result in the expression of certain physical traits (i.e. hair color, eye color, skin color, etc.) or the development of genetic disorders and predisposition to disease.
Personalized, or “precision” medicine is a new approach to diagnostics, treatment, and prevention that takes into account an individual person’s unique genetic profile when developing a medical course of action. Our current “one-size fits all” medical model does not benefit individual patients - because of genetic differences, the same treatment plan may not be as effective for two different patients with the same condition.
Monoclonal antibodies (mAbs), a promising new generation of drug therapy, allows medical experts to treat certain cancers, pathogens (bacteria, virus, fungi, or parasite), and toxins by targeting the individual cancerous cells/pathogen/toxin molecules.
Monoclonal antibodies are synthetic antibodies (a y-shaped protein that neutralizes pathogens and is naturally produced by immune B cells) that scientists design to neutralize specific cellular targets. The term, ‘monoclonal,’ indicates that these antibodies were derived from a single clone of a specific B cell, a.k.a. they only recognize and target one type of antigen (the part of the pathogen or unhealthy cell that the immune system recognizes as foreign).
This technology is particularly helpful in cancer therapy, because it is rare to find identical tumor cells. Therefore, scientists can design monoclonal antibodies that target individual tumor cell types. This differs from our current method of treating cancer patients with chemotherapy or radiation. Both chemotherapy and radiation have serious side effects; they can destroy immune and healthy cells while neutralizing the malignant cells. This is very detrimental to the health of the patient.
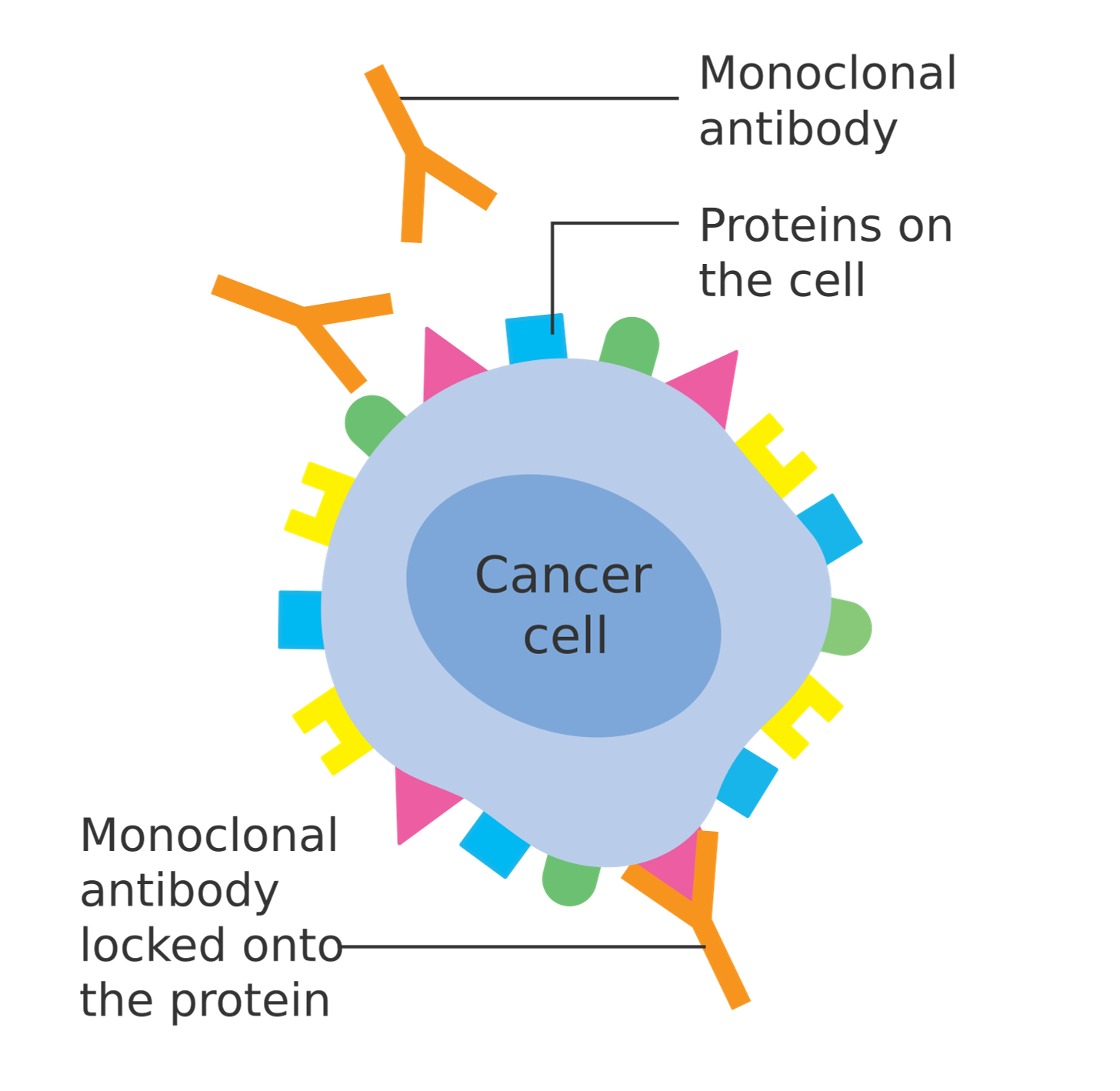
So how do they work?
The mAbs are designed to either neutralize a specific target or increase the patient’s immune response. For example, one type of mAb recognizes and attaches to a specific cellular receptor (HER2) on HER2+ breast and stomach cancer cells. This attachment prevents the cancerous cells from multiplying and spreading. The image above illustrates this process. Another type of mAb allows immune cells to recognize certain types of malignant cells and initiate an immune response.
mAbs need to be used with care; an increased immune response could result in autoimmunity (the body attacks itself).
Recently, the FDA granted casirivimab/imdevimab, a monoclonal antibody cocktail used to treat mild-moderate Covid-19 cases, emergency use authorization. This cocktail targets the SARS-CoV-2 spike protein while preventing mutational escape. Escape mutations are mutations in the antigen protein that prevents antibody recognition and neutralization, in this case the spike protein. In other words, the pathogen escapes detection.
Monoclonal antibodies are the new era of personalized medicine. They allow medical experts to target individual cell types, microorganisms, and viruses, instead of broadly treating a condition which could cause collateral damage.
Keywords:
Personalized “precision” medicine: Health-care designed to benefit the individual patient, often by using genetic profiling.
Monoclonal antibodies (mAbs): Synthetic antibodies derived from a single B cell clone. Made using mice and yeast models.
Escape mutation: Mutations in the antigen protein that prevents antibody recognition and neutralization.
Additional Readings:
FDA Monoclonal Antibody Press Release
Cancer Research - Monoclonal Antibodies